Ultracet Tablets
ULTRACET® tablets are indicated for the short-term (five days or less) management of acute pain.
Ingredients
The two active ingredients are tramadol hydrochloride, a centrally acting synthetic opioid analgesic, and acetaminophen, a non-opiate, non-salicylate analgesic and fever reducer.
- Together they potentiate each other.
Uses
It is used for the relief of moderate to severe acute pain. It comes in two distinct formulations – immediate-release (IR) and extended-release (ER).
Table
| Property | Ultracet IR |
| Description | Opioid pain medication |
| Classification | Narcotic analgesic |
| Administration | Oral, with or without food |
| Color | Yellow |
| Shape | Round |
| Size | Not specifically defined, typically standard tablet size |
| Composition | 37.5 mg Tramadol, 325 mg Acetaminophen |
| Markings | “ORTHO-MCNEIL” and “650” |
| Elimination Time | ~6 hours |
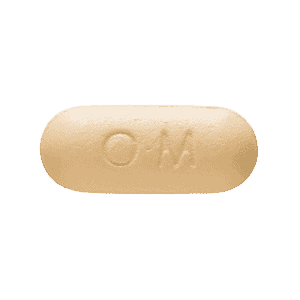
Appearance and Composition
Ultracet IR are typically yellow, round, and film-coated with an imprint of “ORTHO-MCNEIL” on one side and “650” on the other side. The two active ingredients in each are:
- Tramadol Hydrochloride 37.5 mg
- Acetaminophen 375 mg
Pharmacokinetics
It is rapidly absorbed after oral administration, with it demonstrating a bioavailability of about 75%. Acetaminophen is almost completely absorbed, demonstrating a bioavailability of 80-100%.
The peak plasma concentration is achieved within two hours after administration for both components. Both drugs cross the blood-brain barrier, exerting their effects centrally.
Half-life and Elimination
The elimination half-life is approximately six hours. Both medicines are metabolized in the liver and excreted through the kidneys.
Abuse
Due to its opioid content, there is a very high risk of misuse. It has been classified as a Schedule IV controlled substance by the DEA, meaning it has a low potential for abuse relative to substances in Schedule III. Nonetheless, misuse can lead to addiction, overdose, or death, especially in individuals with a history of substance abuse or mental health disorders.
Overdosing Concerns
Overdose symptoms may include respiratory depression, sleepiness leading to coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, in some cases, bradycardia, hypotension, partial or complete airway obstruction, atypical snoring, and death. Acetaminophen overdose can cause acute liver failure, at times resulting in liver transplant and death. Therefore, patients should be instructed to take them only as prescribed.
Important
Please note that this article provides general information only. It should not be used as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
image courtesy of drugs.com

